Search
- Page Path
- HOME > Search
Image of Interest
- Thyroid
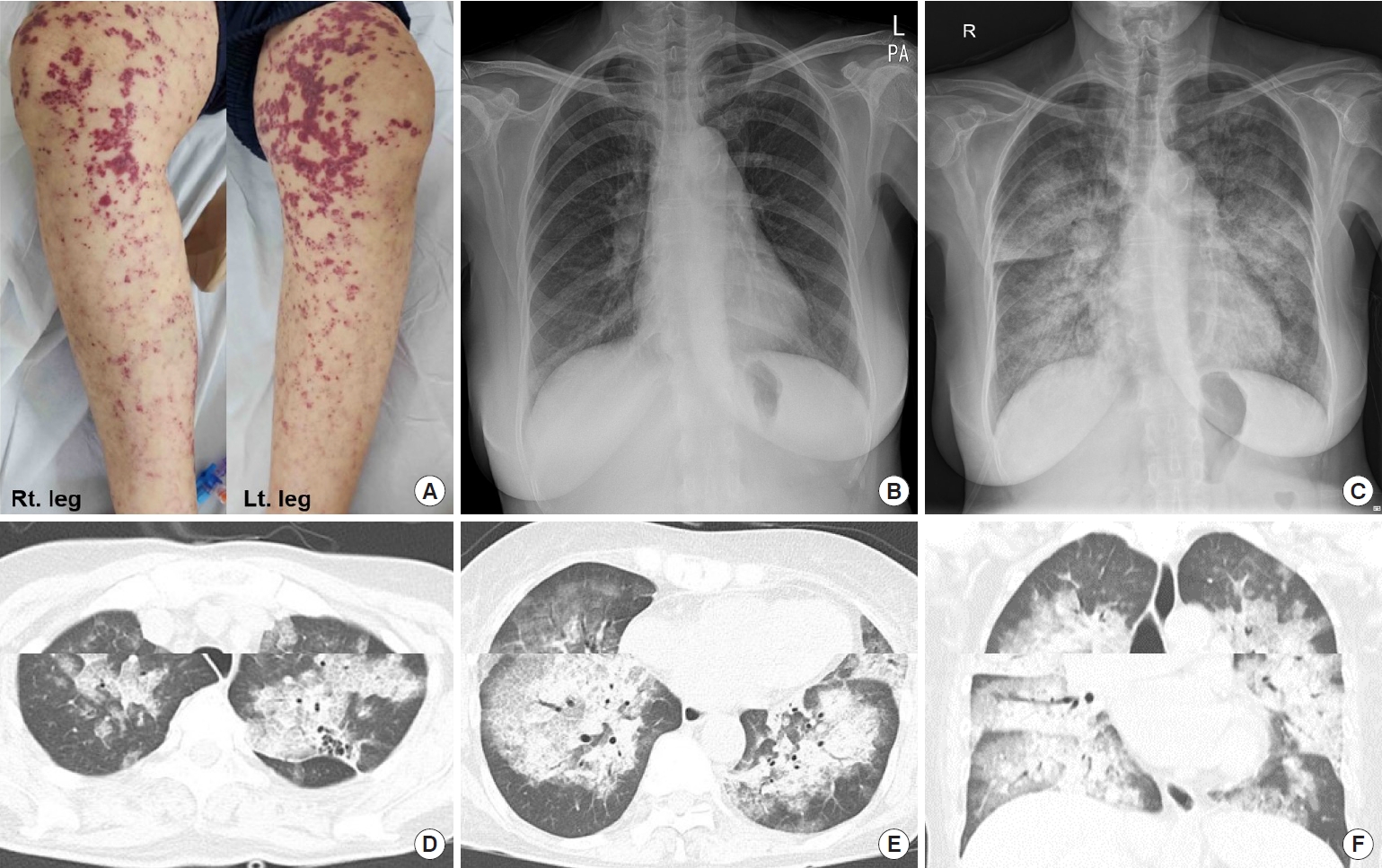
- Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Positive Vasculitis and Agranulocytosis: A Rare Case with Life-Threatening Multiple Systemic Manifestations
- Da Hyun Kang, Mi-Kyung Song, Sang-Hyeon Ju, Song-I Lee, Yea Eun Kang
- Endocrinol Metab. 2023;38(2):282-284. Published online March 13, 2023
- DOI: https://doi.org/10.3803/EnM.2022.1643
- 1,248 View
- 94 Download
- 1 Crossref

Review Article
- Thyroid
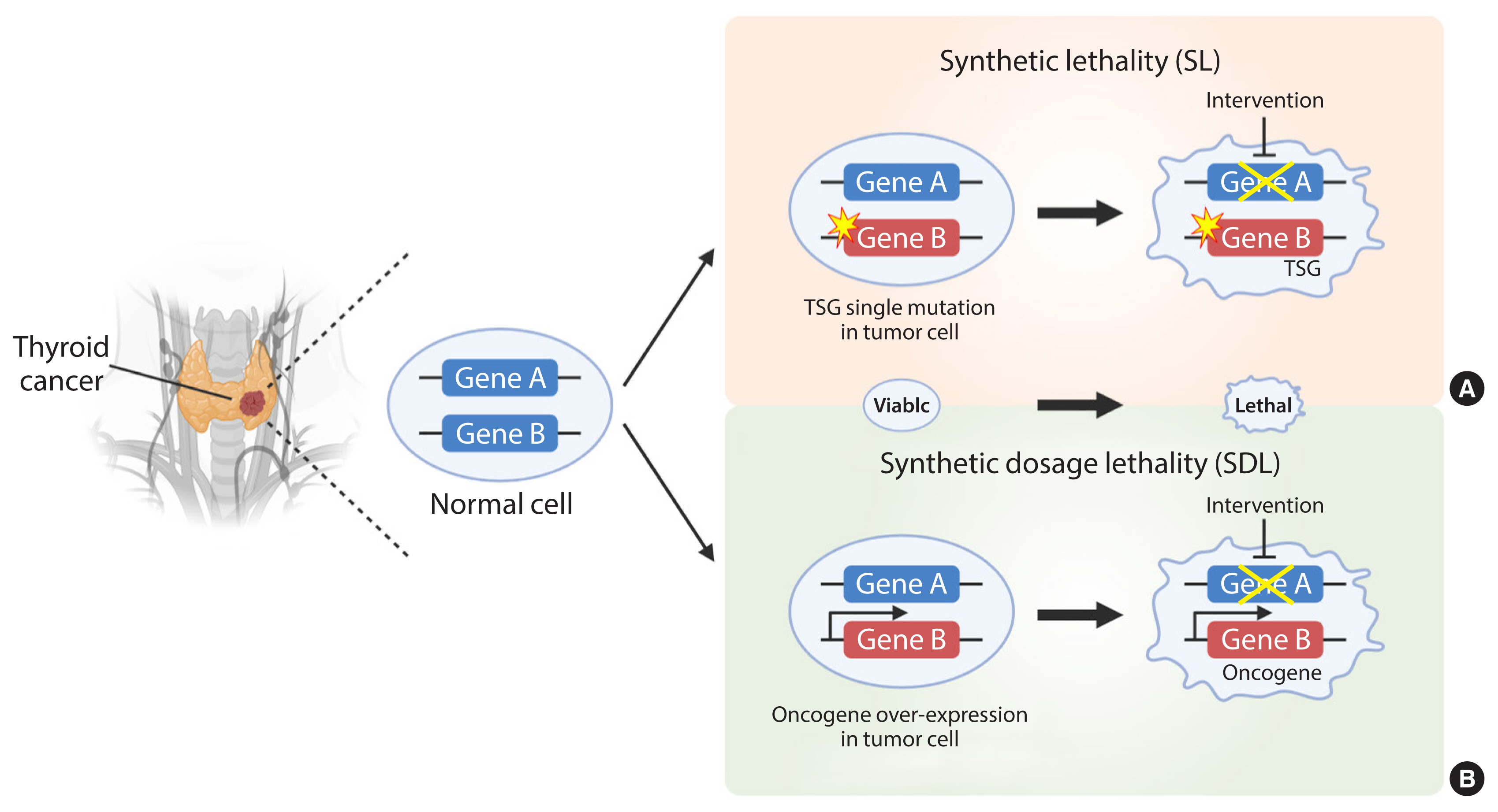
- Development of Metabolic Synthetic Lethality and Its Implications for Thyroid Cancer
- Sang-Hyeon Ju, Seong Eun Lee, Yea Eun Kang, Minho Shong
- Endocrinol Metab. 2022;37(1):53-61. Published online February 28, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1402
- 3,480 View
- 186 Download
- 1 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Cancer therapies targeting genetic alterations are a topic of great interest in the field of thyroid cancer, which frequently harbors mutations in the RAS, RAF, and RET genes. Unfortunately, U.S. Food and Drug Administration-approved BRAF inhibitors have relatively low therapeutic efficacy against BRAF-mutant thyroid cancer; in addition, the cancer often acquires drug resistance, which prevents effective treatment. Recent advances in genomics and transcriptomics are leading to a more complete picture of the range of mutations, both driver and messenger, present in thyroid cancer. Furthermore, our understanding of cancer suggests that oncogenic mutations drive tumorigenesis and induce rewiring of cancer cell metabolism, which promotes survival of mutated cells. Synthetic lethality (SL) is a method of neutralizing mutated genes that were previously considered untargetable by traditional genotype-targeted treatments. Because these metabolic events are specific to cancer cells, we have the opportunity to develop new therapies that target tumor cells specifically without affecting healthy tissue. Here, we describe developments in metabolism-based cancer therapy, focusing on the concept of metabolic SL in thyroid cancer. Finally, we discuss the essential implications of metabolic reprogramming and its role in the future direction of SL for thyroid cancer.
-
Citations
Citations to this article as recorded by- Toward Systems-Level Metabolic Analysis in Endocrine Disorders and Cancer
Aliya Lakhani, Da Hyun Kang, Yea Eun Kang, Junyoung O. Park
Endocrinology and Metabolism.2023; 38(6): 619. CrossRef - The Role of De novo Serine Biosynthesis from Glucose in Papillary Thyroid Cancer
Seong Eun Lee, Na Rae Choi, Jin-Man Kim, Mi Ae Lim, Bon Seok Koo, Yea Eun Kang
International Journal of Thyroidology.2023; 16(2): 175. CrossRef
- Toward Systems-Level Metabolic Analysis in Endocrine Disorders and Cancer


 KES
KES

 First
First Prev
Prev



